On Mar 28th, 2019, China CDE announced a new batch of Reference Listed Drugs on its website for public opinion from Mar 28th to April 15th. It’s the 21st batch of RLD list published officially since China initiated the industry-wide generic drug consistency evaluation, and it’s the first time injection RLD being included. Among the total 327 RLDs the number of injectable dosage form is 224. After the public announcement, this new batch of RLD will be published by China NMPA. The industry consider this as a signal that the China authority will drive forward the consistency evaluation of injections rapidly in near future.
The other difference of this latest batch of RLD comparing to the former twenty batches is, this batch of RLD is selected based on the newly published NMPA Notice No.25 2019. This notice will replace the original SFDA Notice No.99 2016, and mainly stipulates the procedure how the RLD for chemical generics will be selected and determined.
The RLD selection criteria in NMPA Notice No.25 2019 is described as:
- The preference sequence of originator drug: 1) originator drug approved in China; 2) the officially confirmed drug produced in China by the originator company itself or after technology transformation; 3) originator drug not approved in China yet;
- If the originator drug production is shut down or there is quality issue of the originator drug which makes the originator drug not proper to be a RLD, the generic drug which has granted market approval and identified as RLD in USA, Japan or EU countries is acceptable. Such criteria is also applicable for those drugs produced in China by the originator company itself or after technology transformation;
- Other drugs have been evaluated by China NMPA about its safety, effectiveness and quality-control.
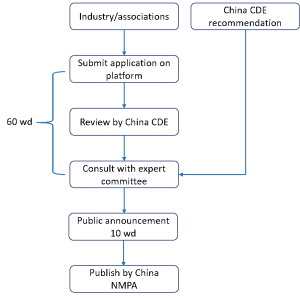
The procedure about how the industries and associations can participate in the RLD selection through the RLD selection application platform is described below:
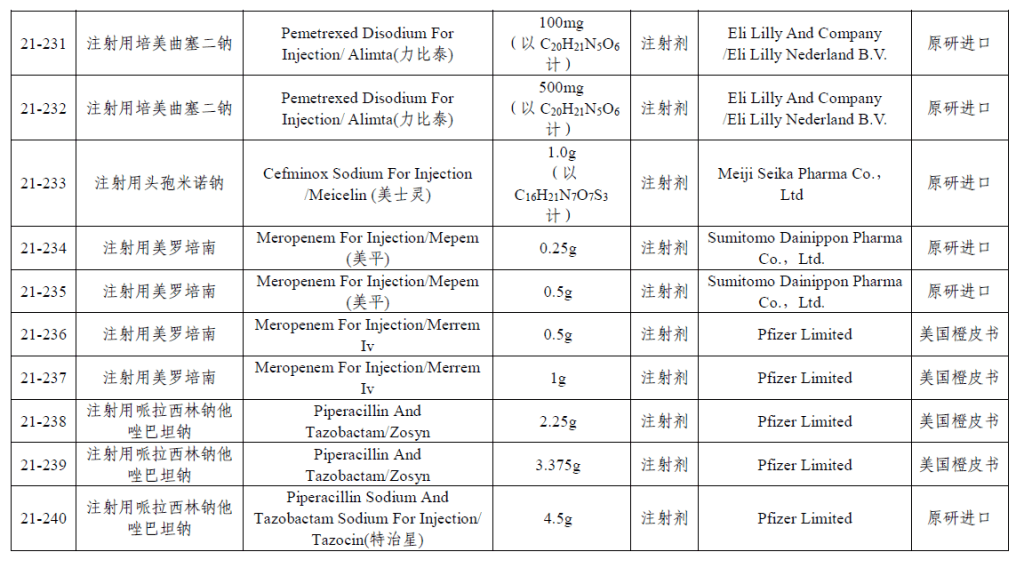
The 21st batch of chemical generic RLD list (part of list)