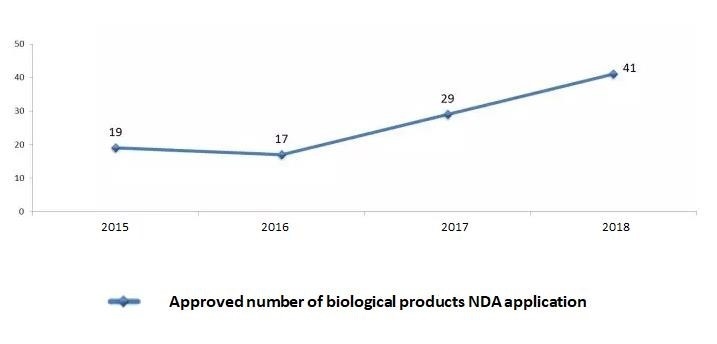
On 2nd July, 2019, China CDE-NMPA published the 2018 working Report, summarizing the drug review and approval statistic data in the past year.
See below for the details.
5- Statistic of Biological Products – Overall
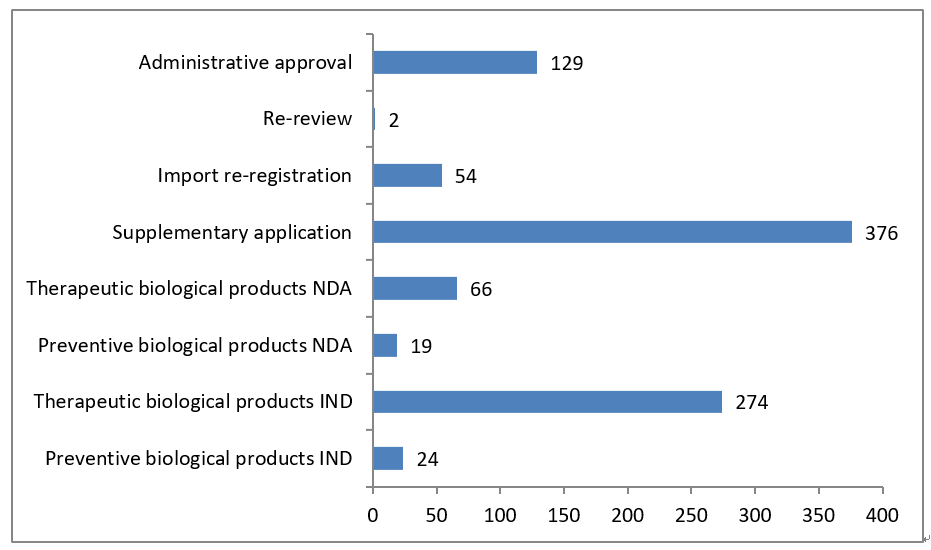
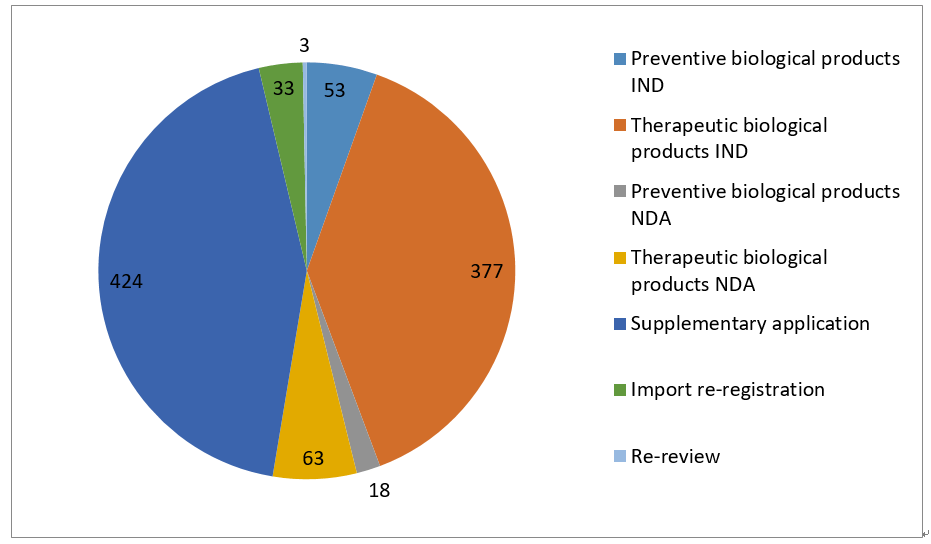
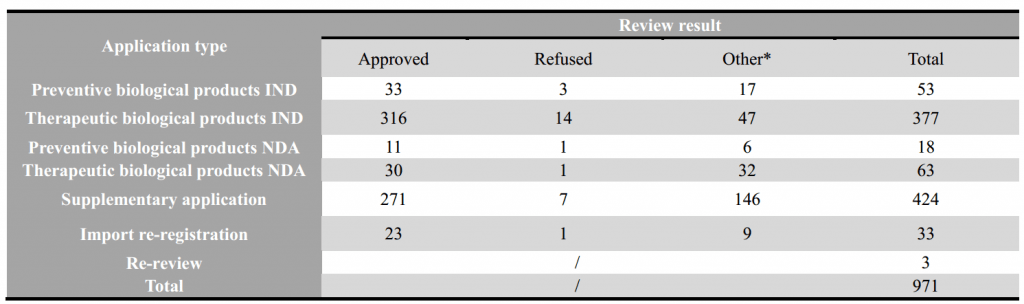
Note: “other” refers: 1) the applicant withdrawal the application, 2) Supplementary information is required from applicant after the review, 3) the application directly submitted to Drug Registration Management Bureau of NMPA instead of CDE, 4) the combination application of medical device, 5) API/excipient/package material application for linked review procedure.
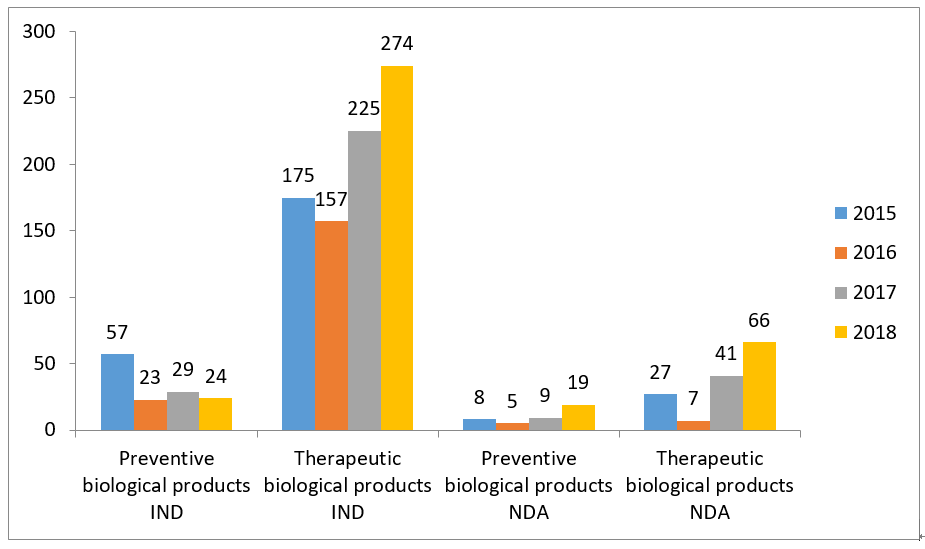
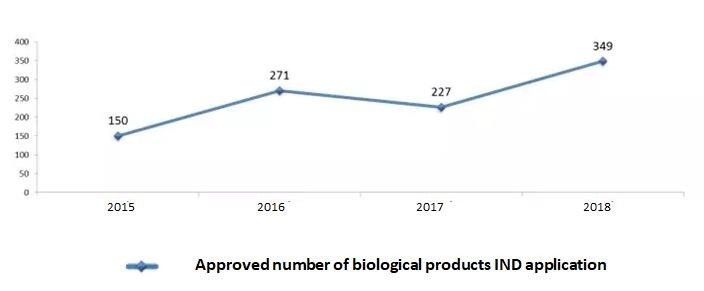
6- Statistic of Biological Products -IND
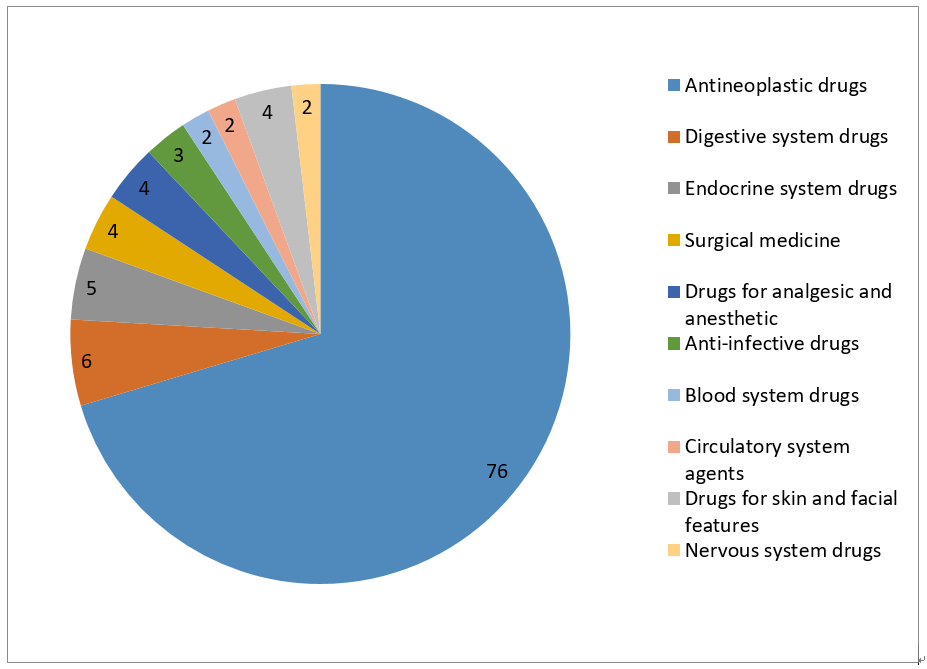
7- Statistic of Biological Products -NDA