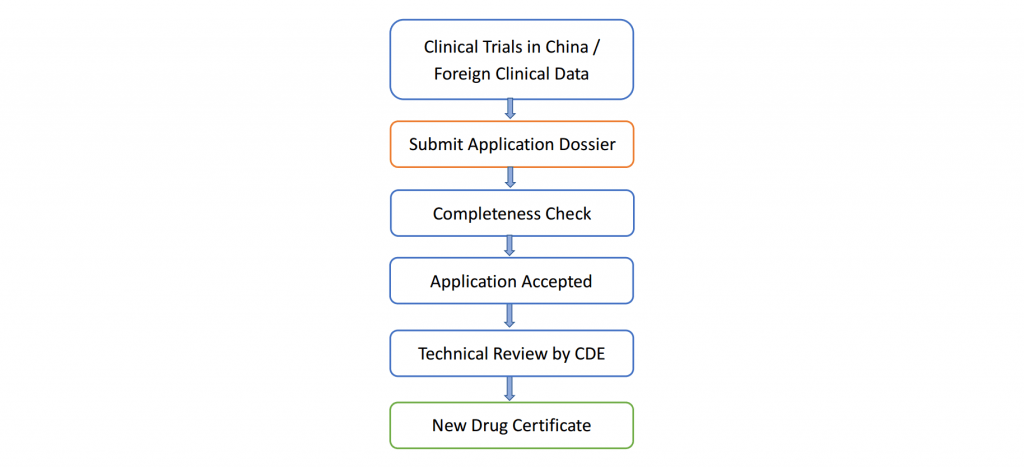
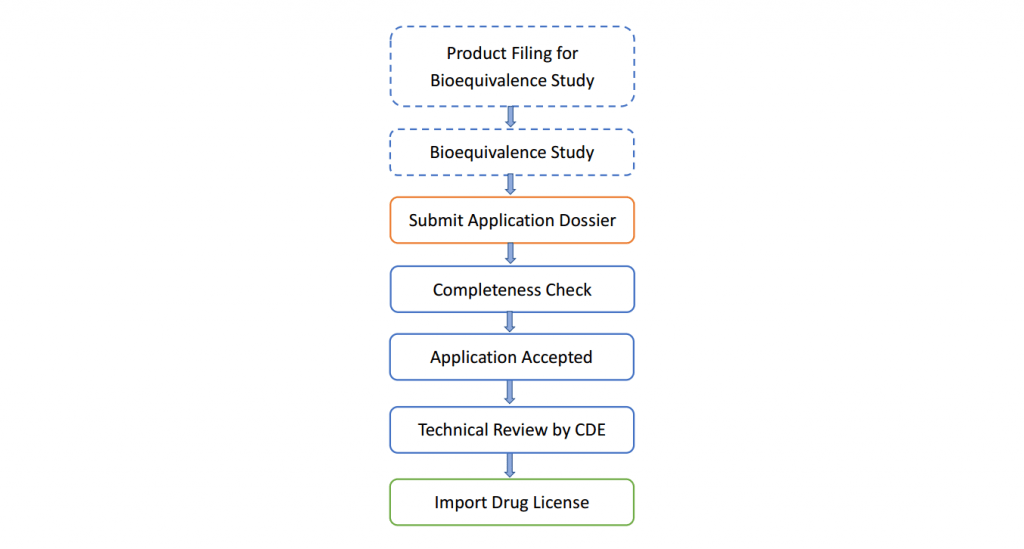
- Workflow of Pre-IND/IND Registration
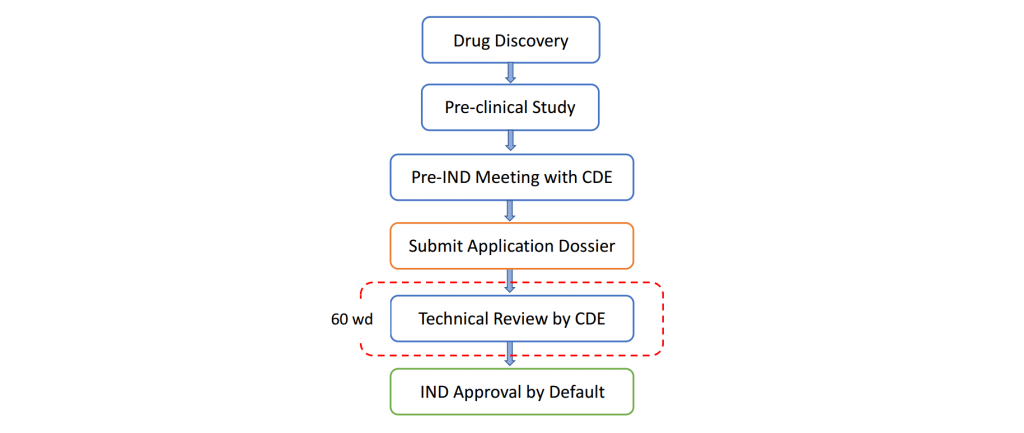
According to the regulatory requirements for drug registrations, such as the Drug Administration Law of the People’s Republic of China (2019 Revision) and the Provisions for Drug Registration (2020 SAMR Decree No. 27), an applicant shall complete the Chemistry, Manufacturing and Controls (CMC) study before applying for an IND registration. The results of quality study, stability test, manufacturing process study and impurity control should be delivered as the scientific basis of clinical trials and provided in the application dossier for CMC review of pre-IND registration. In order to identify the purposes of clinical trials and evaluate the expected benefits and possible risks of clinical trials for the participants and public health, the Center for Drug Evaluation (CDE) of National Medical Products Administration (NMPA) will not only conduct a CMC review but also arrange pre-IND communications or meetings with the applicant. The CDE reviewers will discuss and select a clinical trial strategy which ultimately meets the scientific as well as ethical requirements. After the meeting, the result of discussion together with other application materials should be submitted to CDE by the IND applicant. CDE will conduct a technical review of the application dossier within 60 workdays. If the dossier is reviewed to be sufficient to fulfill the regulatory requirements for clinical trials, i.e. sufficient evidence for efficacy & safety of the drug and no technical or completeness deficiency of the dossier, an IND approval will be issued by default and the applicant will be allowed to conduct clinical trials.
The workflow of pre-IND/IND registration in China is as follows (Fig. 1):